The term “ergogenic” stems from the Greek roots – “Ergon” and “genes,” meaning “work” and “born,” respectively. Any means of enhancing energy production or utilization may be described as an ergogenic aid.1 Ergogenic aids have classically been classified into five categories: mechanical, psychological, physiologic, pharmacologic, and nutritional.2 The present use of the term “ergogenic aid” usually revolves around the physiologic, pharmacologic, and nutritional categories.
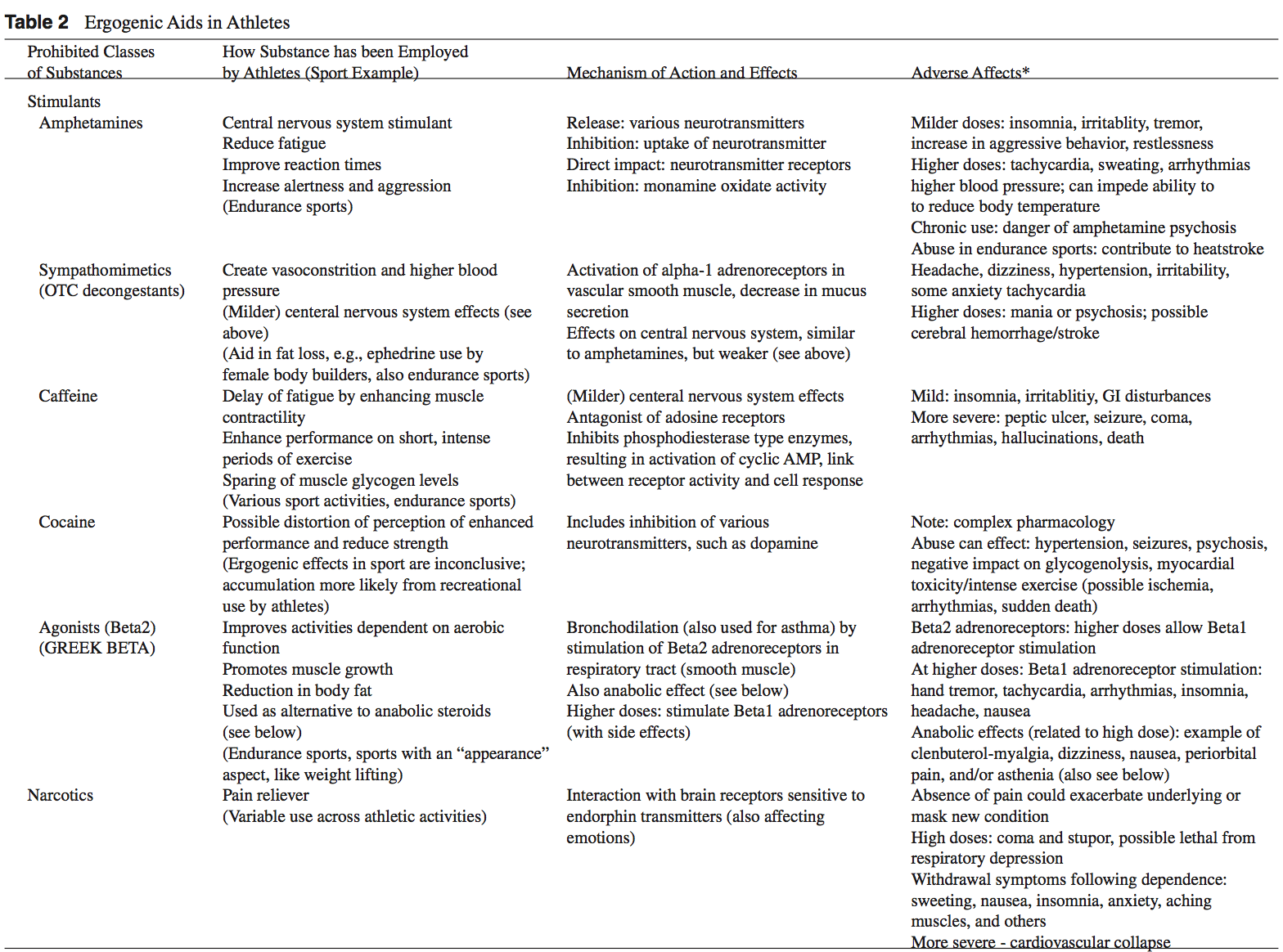
While ergogenic aids have been linked to athletic “doping,” the terms are not synonymous. Doping is a term used by the International Olympic Committee (IOC) to describe the administration or use of a substance by a competing athlete with the sole intention of increasing in an artificial and unfair manner his or her performance in competition.3 Not all ergogenic aids are banned by the IOC. A partial listing of substances banned by the United States Olympic Committee is found in Table 1.2,3 Table 2 provides a list of commonly used athletic ergogenic aids.
Ergogenic Aids:

 Anabolic-Androgenic Steroids
Anabolic-Androgenic Steroids
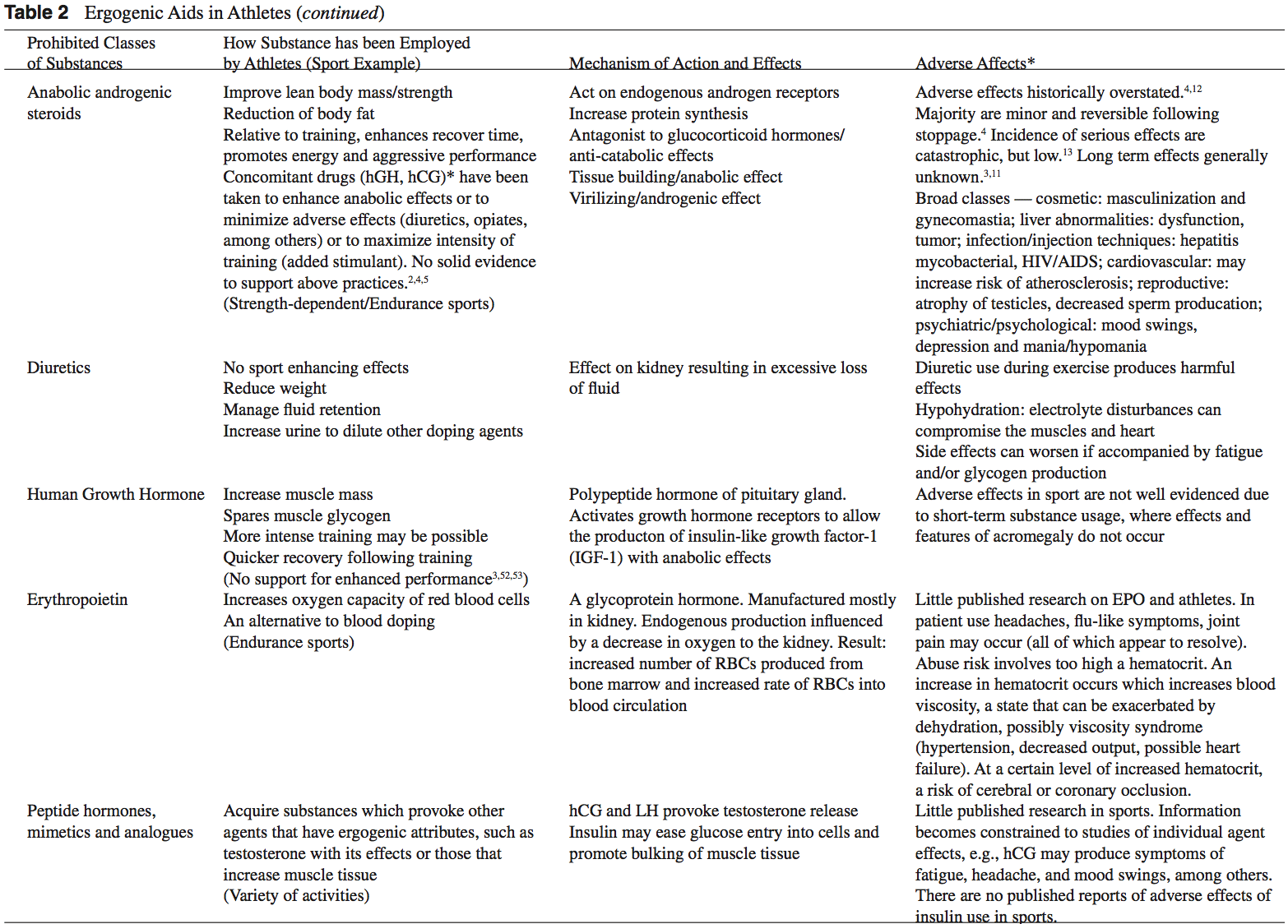
 Anabolic-androgenic steroids (AAS) are testosterone derivatives that exert anabolic (tissue building) and androgenic (masculinizing) influences on the body.3 Since the discovery of the chemical structure of testosterone in 1935, attempts to separate the anabolic and androgenic effects of AAS have been unsuccessful.3 Athletes have been using AAS since the 1940s in efforts to improve their performance.2 Concerned with widespread abuse of AAS among athletes, the IOC banned AAS use in the early 1960s.2 The Anabolic Steroids Control Act was legalized in 1990, making it a felony to possess or distribute AAS for non-medical purposes in the United States.3,4 Oral, parenteral, transdermal, and intra-nasal forms of AAS are available. The vast majority of AAS used by athletes is thought to be obtained on the “black market,” as only an estimated 10% to 15% of AAS used by athletes for performance enhancement are obtained by prescription.3
Anabolic-androgenic steroids (AAS) are testosterone derivatives that exert anabolic (tissue building) and androgenic (masculinizing) influences on the body.3 Since the discovery of the chemical structure of testosterone in 1935, attempts to separate the anabolic and androgenic effects of AAS have been unsuccessful.3 Athletes have been using AAS since the 1940s in efforts to improve their performance.2 Concerned with widespread abuse of AAS among athletes, the IOC banned AAS use in the early 1960s.2 The Anabolic Steroids Control Act was legalized in 1990, making it a felony to possess or distribute AAS for non-medical purposes in the United States.3,4 Oral, parenteral, transdermal, and intra-nasal forms of AAS are available. The vast majority of AAS used by athletes is thought to be obtained on the “black market,” as only an estimated 10% to 15% of AAS used by athletes for performance enhancement are obtained by prescription.3
AAS are believed to exert their main effect by increasing anabolic processes and inhibiting catabolic processes via specific receptor mediated responses within the target cells.5 Effects of AAS include: the anabolic build-up of muscle mass, the androgenic development of secondary male sexual characteristics, an anti-catabolic reversal of cortisol’s action, and a direct psychological effect thought to allow a more intense and sustained workout.2,5-8 Early studies of AAS and athletes produced mixed results.5,6 More recent reviews support the notions that AAS can provide significant increases in muscle mass and strength in athletes.2,5,6 In order to maximize the effects of AAS on strength and power athletes, an adequate diet and exercise regimen is needed.5 There seems to be little advantage gained while using AAS in the untrained individual.5,9 Benefits obtained from AAS are more established in strength-dependent sports. Data supporting increased aerobic capacity and improved endurance with AAS use is limited and inconclusive.4 AAS effect on endurance sports is currently an area of great interest given the large number of endurance athletes who still use AAS.4,10
An intricate terminology describing the dosing practices of athletes has evolved. Athletes will commonly use AAS over 6 to 12 week “cycles.”4 “Pyramiding” describes a gradual escalation in the dose of AAS taken over a cycle.2,11 “Stacking” involves the use of more than one AAS, usually with staggered cycles of the individual drugs.2-4 An “array” describes the practice of using other drugs to counteract side effects or enhance the effects of AAS.3 The practices of cycling, pyramiding, and stacking are used by athletes in an attempt to minimize the negative effects of AAS while maximizing the desired enhancements.2,4 At the current time, no solid scientific support exists for these practices.2,4,5
The adverse effects attributed to AAS abuse have been historically overstated.4,12 The majority of AAS side effects are considered minor and reversible following the cessation of use.4 While the incidence of serious side effects from AAS use has been low, devastating consequences have been reported.13 Documented fatalities from myocardial infarc- tion, stroke, and hepatocarcinoma have been attributed to AAS use.2,3 The long-term effects of AAS use are generally unknown.3,11
Dehydroepiandrosterone (DHEA)
 Dehydroepiandrosterone (DHEA) is a precursor to testos- terone produced primarily in the adrenal glands.4,14 Natural sources of DHEA include wild yams. The FDA banned sale of DHEA in 1996 due to insuf cient evidence of safety and value; however, DHEA remains a legal and popular item sold as a nutritional supplement.14,15
Dehydroepiandrosterone (DHEA) is a precursor to testos- terone produced primarily in the adrenal glands.4,14 Natural sources of DHEA include wild yams. The FDA banned sale of DHEA in 1996 due to insuf cient evidence of safety and value; however, DHEA remains a legal and popular item sold as a nutritional supplement.14,15
The mechanism of action of DHEA is poorly understood but most likely revolves around the conversion of DHEA to testosterone in peripheral tissues.4,14 Preliminary studies suggest that DHEA may have a broad range of clinical uses including anti-Alzheimer and anti-Parkinson capabilities, however randomized, double-blinded clinical studies are lacking.5
DHEA is a pre-cursor to testosterone and theoretically may enhance athletic performance in a manner similar to AAS. Investigations of DHEA use and athletic performance are scarce.14 Existing studies do not support a significant increase in lean body mass, strength, or testosterone levels with the use of DHEA in athletes.14,16-18
Long-term side effects of DHEA use are currently un- known but are probably similar to those associated with AAS use.6,14
Androstenedione
 Androstenedione is a testosterone pre-cursor produced in the adrenal glands and gonads. Several professional athletes have used this substance, bringing it to national attention.2 Androstenedione is found naturally in the pollen of Scottish pine trees.19
Androstenedione is a testosterone pre-cursor produced in the adrenal glands and gonads. Several professional athletes have used this substance, bringing it to national attention.2 Androstenedione is found naturally in the pollen of Scottish pine trees.19
Similar to DHEA, the mechanism of action and side ef- fects attributed to androstenedione are poorly understood and thought to be related to the conversion of androstenedione to testosterone in the peripheral tissues.5
Despite manufacturers’ claims to the contrary, there is little scientific evidence of the purported ergogenic aid effects of androstenedione.2,5,16,20 Recently concerns have grown over the unfavorable alterations in blood lipid and coronary heart disease profiles seen in men using androstenedione as an ergogenic aid.2,20,21
Dietary Supplements
 The increased visibility of ergogenic aids in the last de- cade has occurred primarily because of the passage of the United States Dietary Supplement Health and Education Act (DSHEA) of 1994.22 Certain vitamins, minerals, amino acids, herbs, and other botanical preparations can be classified as a “dietary supplement” under the DSHEA guidelines. Dietary supplements, as a result of DSHEA, are no longer under the direct regulatory control of the FDA. In fact, substances sold as a dietary supplement do not require FDA evaluation for safety or efficacy, and do not have to meet quality control standards expected of approved drugs.5 The content and purity of dietary supplements are not regulated and can vary widely.5,23 Since androstenedione and DHEA have been found to occur naturally in plant sources, these testosterone precursors can be labeled as “dietary supplements” and sold legally over-the-counter.
The increased visibility of ergogenic aids in the last de- cade has occurred primarily because of the passage of the United States Dietary Supplement Health and Education Act (DSHEA) of 1994.22 Certain vitamins, minerals, amino acids, herbs, and other botanical preparations can be classified as a “dietary supplement” under the DSHEA guidelines. Dietary supplements, as a result of DSHEA, are no longer under the direct regulatory control of the FDA. In fact, substances sold as a dietary supplement do not require FDA evaluation for safety or efficacy, and do not have to meet quality control standards expected of approved drugs.5 The content and purity of dietary supplements are not regulated and can vary widely.5,23 Since androstenedione and DHEA have been found to occur naturally in plant sources, these testosterone precursors can be labeled as “dietary supplements” and sold legally over-the-counter.
Ephedra
 Dietary supplements containing Chinese ephedra, also known as Mahaung, are marketed as performance enhancers and weight-loss aids.24 Ephedra species of herb have been used for over 5,000 years for respiratory ailments.25 Currently, ephedrine alkaloids are found in hundreds of prescriptions and over-the-counter products, such as antihistamines, decongestants, and appetite suppressants.24-26 Ephedra and related ephedrine alkaloids are sympathomimetic agents that mimic epinephrine effects.
Dietary supplements containing Chinese ephedra, also known as Mahaung, are marketed as performance enhancers and weight-loss aids.24 Ephedra species of herb have been used for over 5,000 years for respiratory ailments.25 Currently, ephedrine alkaloids are found in hundreds of prescriptions and over-the-counter products, such as antihistamines, decongestants, and appetite suppressants.24-26 Ephedra and related ephedrine alkaloids are sympathomimetic agents that mimic epinephrine effects.
Multiple studies of isolated ephedrine alkaloids have shown no significant enhancement of power or endurance at dosages considered to be safe.24,27-31 In contrast, the combination of caffeine with ephedrine has been associated with improvements in performance and may promote metabolic effects that are conducive to body fat loss.26,32
The actual content of ephedra alkaloids in 20 ephedra- containing dietary supplements was studied using high- performance liquid chromatography.33 Ten of the twenty supplements exhibited marked discrepancies between the label claim for ephedra content and the actual alkaloid content. Between 1995 and 1997, 926 cases of possible Mahuang toxicity were reported to the Food and Drug Ad- ministration.34 A temporal relationship between Mahuang use and severe complications including stroke, myocardial infarction, and sudden death was established in 37 of the 926 cases. In 36 of these 37 cases, the Mahuang use was reported to be within the manufacturers’ dosing guidelines.
Ephedra and related ephedrine alkaloids are currently banned by the U.S.O.C. and cannot be recommended for general use given their association with potentially life- threatening side effects.2,34
Creatine
 Creatine use in athletes has grown as a result of a 1992 study that showed that creatine supplementation produced a 20% increase in skeletal muscle creatine concentration.2,35 In the phosphorylated form, creatine serves as an energy substrate that contributes to adenosine triphosphate (ATP) re-synthesis during high-intensity exercise.36 Creatine re- mains popular with power and resistance athletes as it is thought to produce increases in strength, muscle mass, and to delay fatigue.2,14,36
Creatine use in athletes has grown as a result of a 1992 study that showed that creatine supplementation produced a 20% increase in skeletal muscle creatine concentration.2,35 In the phosphorylated form, creatine serves as an energy substrate that contributes to adenosine triphosphate (ATP) re-synthesis during high-intensity exercise.36 Creatine re- mains popular with power and resistance athletes as it is thought to produce increases in strength, muscle mass, and to delay fatigue.2,14,36
Creatine is synthesized from amino acids primarily in the liver, pancreas, and kidney and is excreted by the kidneys. Creatine is found in skeletal muscle, cardiac muscle, brain, retinal, and testicular tissues.2,37 The interest in creatine as an ergogenic aid revolves around its ability to participate as an energy substrate for muscle contraction.14 Creatine, which easily binds phosphorus, can act as a substrate to donate phosphorus for the formation of ATP. Furthermore, creatine-phosphate (PCr) can help buffer lactic acid because hydrogen ions are used when ATP is regenerated.14,36,38 This role of creatine in exercise is governed by the following reaction:
PCr + ADP (adenosine diphosphate) ↔ Creatine + ATP.(metzl) Creatine kinase
Normally PCr stores deplete within 10 seconds of short, high-intensity exercise.14,39 Increasing the level of PCr in skeletal muscle, in theory, should result in the ability to sustain high-power output longer and lead to a greater re-synthesis of PCr after exercise.14 The beneficial effects of creatine in response to resistance training are most likely mediated by the following sequence: increased muscle creatine concentration, increased training intensity, which lead to an enhanced physiologic adaptation to training with increased muscle mass and strength.36
Studies evaluating the effectiveness of creatine as an er- gogenic aid are mixed.2,36,40 Multiple reports do conclude that short-term creatine supplementation signi cantly enhances the ability to maintain muscular force and power output dur- ing high-intensity exercise.2,36,41,42 Data on results of creatine supplementation with highly trained athletes is inconclusive. While some papers report improvements with creatine use in highly trained individuals with regards to high-intensity exercise, many show no improvements.2,36,43
Most investigators agree that creatine supplementation does not seem to enhance aerobic-oriented activities.2,36,44
Human muscle is thought to have a maximum concen- tration of creatine that it can hold.14,45 There appears to be no additional bene ts of increasing creatine supplementa- tion above this storage capacity of muscle as the excess is simply excreted by the kidneys.2,46 Humans have differing baseline levels of muscle creatine.14 Accordingly, athletes with lower baseline levels of creatine may be more sensi- tive to creatine supplementation than those with a relatively higher baseline creatine level.14,36 The terms “responder” and “nonresponder” have been used to describe two groups of athletes: those with relatively low baseline creatine levels that may show signi cant performance enhancement with creatine supplementation, and those with high baseline creatine levels that do not show marked improvements with creatine supplementation.14,36,47 These differences in creatine concentrations are thought to play a signi cant role in the varied results on performance found in the literature examin- ing creatine supplementation.14
Reported side effects from creatine use have been scarce.2,14 The major reported side effect associated with creatine use is weight gain, which is thought to be primarily a result of water retention.2,14,48 Some reported longer-term side effects include dehydration, muscle cramping, nausea, and seizures.2,49 Given the relative lack of studies, caution still remains about the long-term effects of creatine usage.14 As creatine use among younger athletes continues to increase, concern is growing over the lack of studies that examine the possible side effects speci c to this age group.14,38
Human Growth Hormone
 Human growth hormone (hGH) is a polypeptide produced in the anterior pituitary gland. After its release from the pituitary, hGH can exert its effect in all cells of the body via tissue specific receptors. Human growth hormone is shown to promote protein anabolism, carbohydrate tolerance, lipolysis, natriuresis, and bone and connective tissue turnover.4,50
Human growth hormone (hGH) is a polypeptide produced in the anterior pituitary gland. After its release from the pituitary, hGH can exert its effect in all cells of the body via tissue specific receptors. Human growth hormone is shown to promote protein anabolism, carbohydrate tolerance, lipolysis, natriuresis, and bone and connective tissue turnover.4,50
Potential benefits of hGH abuse in athletes revolve around its anabolic effect on the body.4 Human growth hormone is thought to increase muscle mass, and spare muscle glycogen by stimulating lipolysis during exercise.2,3 The popularity of hGH among athletes is furthered by the fact that hGH re- mains extremely difficult to detect by current drug screening processes.3,51 Human growth hormone may be particularly attractive to female athletes as the virilization side effects associated with AAS use are not thought to occur with hGH.4
There are no studies that demonstrate signi cant increases in athletic performance with the use of hGH.3,52,53 Neither human or animal studies show any signi cant strength gains with supplemental hGH use in non-de cient individuals.4 The abuse of hGH is thought to be increasing despite the lack of scienti c evidence linking hGH to improved athlete performance.3,52 A survey of high school males revealed that as many as 5% reported past or present use of hGH.54 The purity of hGH abused by athletes may be poor as Drug Enforcement Agency estimates project that up to 30% to 50% of the hGH products sold are phony.4,55
Adverse effects of exogenous hGH use are extrapolated from the ndings seen in patients with endogenous over- secretion of hGH.2 Adults with high levels of hGH are at risk for the clinical syndrome of acromegaly. Medical complications associated with acromegaly include: diabetes, hypertension, coronary heart disease, cardiomyopathy, men- strual irregularities, and osteoporosis.2,4 High levels of hGH in individuals with open physis may lead to gigantism.2
Erythropoietin (EPO)
 Recombinant EPO (r-EPO) was approved by the FDA for manufacture in 1989 after the EPO gene was cloned in 1985.14 Since its approval, r-EPO has been abused for athletic personal gain as an alternative to blood doping.3,14 Recombinant EPO has largely replaced the practice of blood doping, as r-EPO produces a dose-dependent increase in hematocrit.2 In theory, r-EPO should provide all of the benefits of blood doping without the risks involved in blood transfusion.3
Recombinant EPO (r-EPO) was approved by the FDA for manufacture in 1989 after the EPO gene was cloned in 1985.14 Since its approval, r-EPO has been abused for athletic personal gain as an alternative to blood doping.3,14 Recombinant EPO has largely replaced the practice of blood doping, as r-EPO produces a dose-dependent increase in hematocrit.2 In theory, r-EPO should provide all of the benefits of blood doping without the risks involved in blood transfusion.3
There are few studies evaluating the use of r-EPO in healthy athletes; however, numerous studies have shown a signi cant increase in work capacity due to r-EPO use in patients with renal disease.14 Berglund and Ekblom reported an increased maximal oxygen consumption and increased time to exhaustion in male athletes after a 6 week trial of r-EPO.56
The risks associated with r-EPO abuse involve the potential for dangerously high hematocrit levels.14 A resulting hyperviscosity syndrome may lead to a decreased cardiac output, hypertension, and potential heart failure.3 Further- more, thrombosis could be manifest as myocardial infarction, pulmonary embolism, or cerebrovascular accidents.2,3 Although the use of r-EPO has been banned by the IOC since 1990, its use is extremely difficult to detect with current drug screening measures.2,14
Antioxidants
 The antioxidant capabilities of certain vitamins are believed by many to counter-act the production of free-radials that occurs during exercise.14 Most of the research to date involves vitamin E, vitamin C, and beta carotene.2 The existing literature does not support the notion that antioxidants have significant ergogenic capabilities.2,14,57 There are currently no recommendations for antioxidant use in athletes that exceeds the normal adult recommended daily allowance (RDA).
The antioxidant capabilities of certain vitamins are believed by many to counter-act the production of free-radials that occurs during exercise.14 Most of the research to date involves vitamin E, vitamin C, and beta carotene.2 The existing literature does not support the notion that antioxidants have significant ergogenic capabilities.2,14,57 There are currently no recommendations for antioxidant use in athletes that exceeds the normal adult recommended daily allowance (RDA).
Beta-Hydroxy-Beta-Methylbutyrate
 Beta-hydroxy-beta-methylbutyrate (HMB) is a metabolite of the branched-chain amino acid leucine. HMB is theorized to inhibit muscle breakdown during strenuous exercise but its exact mechanism of action remains unknown.14,58 Studies show that HMB supplementation may significantly lower serum lactate dehydrogenase (LDH), lower serum creatine phosphokinase (CPK) levels and delay blood lactate accumulation after endurance training compared to placebo.59,60 Furthermore, short-term HMB use has been shown to significantly increase strength gains with resistance-exercised training over placebo in one double-blinded study.61
Beta-hydroxy-beta-methylbutyrate (HMB) is a metabolite of the branched-chain amino acid leucine. HMB is theorized to inhibit muscle breakdown during strenuous exercise but its exact mechanism of action remains unknown.14,58 Studies show that HMB supplementation may significantly lower serum lactate dehydrogenase (LDH), lower serum creatine phosphokinase (CPK) levels and delay blood lactate accumulation after endurance training compared to placebo.59,60 Furthermore, short-term HMB use has been shown to significantly increase strength gains with resistance-exercised training over placebo in one double-blinded study.61
HMB is a relatively new ergogenic aid and published results are considered preliminary.14,58 Although there is evidence for a potential ergogenic aid advantage with HMB use in resistance and endurance training, its use can not be recommended until more studies are performed and potential side effects are elicited.
Caffeine
 Caffeine is a methylxanthine occurring naturally in many species of plants. Caffeine is thought to work through a variety of mechanisms. The central nervous system effect of caffeine is probably the result of adrenergic receptor antagonism.3 Its use by athletes stems from the theory that caffeine may delay fatigue by enhancing skeletal muscle contractility and spare muscle glycogen levels by enhancing fat metabolism.6 Multiple studies have reported an improved endurance time with caffeine use.6,62,63 There is evidence that caffeine use may enhance performance with more intense short-duration exercise as well.2 The caffeine dosages most associated with an ergogenic effect range in the literature from 3 to 9 mg per kilogram of body weight.2,6
Caffeine is a methylxanthine occurring naturally in many species of plants. Caffeine is thought to work through a variety of mechanisms. The central nervous system effect of caffeine is probably the result of adrenergic receptor antagonism.3 Its use by athletes stems from the theory that caffeine may delay fatigue by enhancing skeletal muscle contractility and spare muscle glycogen levels by enhancing fat metabolism.6 Multiple studies have reported an improved endurance time with caffeine use.6,62,63 There is evidence that caffeine use may enhance performance with more intense short-duration exercise as well.2 The caffeine dosages most associated with an ergogenic effect range in the literature from 3 to 9 mg per kilogram of body weight.2,6
Side effects associated with caffeine use include anxiety, diuresis, insomnia, irritability and gastrointestinal discomfort. 2,6 Higher doses of caffeine ingestion can lead to more serious consequences such as cardiac arrhythmia, hallucinations, and even death.2,3
The legal urine level of caffeine for athletes is 12 μg/ml (IOC standards) and 15 μg/ml (National Collegiate Athletics Association standards).6 An athlete would need to drink six to eight cups of coffee in one sitting and be tested within 2 to 3 hours to reach urine levels over the IOC legal limit.3 The amount of caffeine needed to produce ergogenic benefits is potentially far less than that required to exceed the athletic legal limit.3
Ergogenic Aids: Summary
Claims championing exotic substances that produce healing or ergogenic powers have been around for centuries. The competitive, peer-pressured environment enveloping today’s athletes and adolescences makes these groups particularly susceptible to the uproar surrounding the current ergogenic aid market. Presently, it seems that rumor and anecdotal information overwhelms the available scientific data. While there is evidence that some touted ergogenic aids do indeed enhance performance, there are many unanswered questions about product safety, efficacy, and long-term consequences. A working knowledge of specific ergogenic aids is essential for the treating physician in order to best advise patients and athletes as to the possible benefits and risks of any substance they may be using.
By Adam Bernstein, M.D., Jordan Safirstein, M.D., and Jeffrey E. Rosen, M.D.
1. Williams MH: Ergogenic and ergolytic substances. Med Sci
Sports Exerc 24(9 Suppl):S344-S348, 1992.
2. Silver MD: Use of ergogenic aids by athletes. J Am Acad
Orthop Surg 9(1):61-70, 2001.
3. KnoppWD,WangTW,Bach JrBR: Ergogenic drugsin sports.
Clin Sports Med 16(3):375-392, 1997.
4. Sturmi JE, Diorio DJ: Anabolic agents. Clin Sports Med
17(2):261-282, 1998.
5. Blue JG, Lombardo JA: Steroids and steroid-like compounds.
Clin Sports Med 18(3):667-689, 1999.
6. Ahrendt DM: Ergogenic aids: counseling the athlete.Am Fam
Physician 63(5):913-922, 2001.
7. Adolescents and anabolic steroids:A subjectreview.American
Academy of Pediatrics. Committee on Sports Medicine and
Fitness. Pediatrics 99(6):904-908, 1997.
8. Haupt HA: Anabolic steroids and growth hormone. Am J
Sports Med 21(3):468-474, 1993.
9. Kuipers H, et al: Influence of anabolic steroids on body composition,
blood pressure, lipid profile and liver functions in
body builders. Int J Sports Med 12(4):413-418, 1991.
10. Lombardo JA: Medical and performance-enhancing effects
of anabolic steroids. Psychiatr Ann 22:19-23, 1992.
11. YesalisCE,Bahrke MS:Anabolic-androgenic steroids: current
issues. Sports Med 19(5):326-340, 1995.
12. Friedl KE: Effects of anabolic steroids on physical health.
In:Yesalis CE (ed): Anabolic Steroids in Sports and Exercise
(2nd ed). Champaign, IL: Human Kinetics Publishers, Inc.,
2000, pp. 35-48.
13. Bahrke MS, Yesalis CE, Brower KJ: Anabolic-androgenic
steroid abuse and performance-enhancing drugs among adolescents.
Child Adolesc Psychiatr Clin N Am 7(4):821-838,
1998.
14. Stricker PR: Other ergogenic agents. Clin Sports Med
17(2):283-297, 1998.
15. Dehydroepiandrosterone (DHEA). Med Lett Drugs Ther
38(985):91-92, 1996.
16. Wallace MB, et al: Effects of dehydroepiandrosterone vs
androstenedione supplementation in men. Med Sci Sports
Exerc 31(12):1788-1792, 1999.
17. Nestler JE, et al: Dehydroepiandrosterone reduces serum
low density lipoprotein levels and body fat but does not alter
insulin sensitivity in normal men. J Clin Endocrinol Metab
66(1):57-61, 1988.
18. Welle S,Jozefowicz R, Statt M: Failure of dehydroepiandrosterone
to influence energy and protein metabolism in humans.
J Clin Endocrinol Metab 71(5):1259-1264, 1990.
19. Saden-Krehula M, Tajic M, Kolbah D: Testosterone, epitestosterone
and androstenedione in the pollen of Scotch pine
P. silvestris L. Experientia 27(1):108-109, 1971.
20. King DS, et al: Effect of oral androstenedione on serum testosterone
and adaptationsto resistance training in young men:
a randomized controlled trial.JAm MedAssoc 281(21):2020-
2028, 1999.
21. Broeder CE, et al: The Andro Project: physiological and
hormonal influences of androstenedione supplementation in
men 35 to 65 years old participating in a high-intensity resistance
training program.Arch Intern Med 160(20):3093-3104,
2000.
22. Benning JR: Nutrition for exercise and sports performance. In:
Mahan LK (ed): Krause’s Food, Nutrition and Diet Therapy.
Philadephia: W.B. Saunders Co., 2000, pp. 534-557.
23. SkolnickAA: Scientific verdictstill out on DHEA.JAm Med
Assoc 276(17):1365-1367, 1996.
24. Bucci LR: Selected herbals and human exercise performance.
Am J Clin Nutr 72(2 Suppl):624S-636S, 2000.
25. Anonymous: The Ephedras. Lawrence Rev Nat Prod, 1989.
26. DiPasquale M: Stimulants and adaptogens: Part I. Drug Sports
1:2-6, 1992.
27. Sidney KH, Lefcoe NM: The effects of ephedrine on the
physiological and psychological responsesto submaximal and
maximal exercise in man. Med Sci Sports 9(2):95-99, 1977.
28. Bright TP, Sandage Jr BW, Fletcher HP: Selected cardiac and
metabolic responsesto pseudoephedrine with exercise.J Clin
Pharmacol 21(11-12):488-492, 1981.
29. DeMeersman R, Getty D, Schaefer DC: Sympathomimetics
and exercise enhancement: all in the mind? Pharmacol Biochem
Behav 28(3):361-365, 1987.
30. SwainRA, et al: Do pseudoephedrine or phenylpropanolamine
improve maximum oxygen uptake and time to exhaustion?
Clin J Sport Med 7(3):168-173, 1997.
31. Gillies H, et al: Pseudoephedrine is without ergogenic effects
during prolonged exercise. J Appl Physiol 81(6):2611-2617,
1996.
32. Bell DG, Jacobs I, Zamecnik J: Effects of caffeine, ephedrine
and their combination on time to exhaustion during
high-intensity exercise. Eur J Appl Physiol Occup Physiol
77(5):427-433, 1998.
33. Gurley BJ, Gardner SF, Hubbard MA: Content versus label
claims in ephedra-containing dietary supplements. Am J
Health Syst Pharm 57(10):963-969, 2000.
34. Samenuk D, et al: Adverse cardiovascular events temporally
associated with ma huang, an herbal source of ephedrine.
Mayo Clin Proc 77(1):12-16, 2002.
35. Juhn MS: Orla creatine supplementation: Separating fact from
hype. Phys Sportsmed 27:47-56, 1999.
36. Kraemer WJ, Volek JS: Creatine supplementation: Its role in
human performance. Clin Sports Med 18(3):651-666, 1999.
37. Williams MH: The use of nutritional ergogenic aidsin sports:
is it an ethical issue? Int J Sport Nutr 4(2):120-131, 1994.
38. MetzlJD, et al: Creatine use among young athletes. Pediatrics
108(2):421-425, 2001.
39. Spriet LL: Ergogenic aids: recent advances and retreats. In:
Lamb DR, Murray R (eds): Perspectives in Exercise Science
and Sports Medicine. Indianapolis, IN: Benchmark Press,
1998, pp. 185-238.
40. Johnson WA, Landry GL: Nutritional supplements: fact vs.
fiction. Adolesc Med 9(3):501-513, 1998.
41. Williams MH, Branch JD: Creatine supplementation and
exercise performance: an update. J Am Coll Nutr 17(3):216-
234, 1998.
42. Mujika I, Padilla S: Creatine supplementation as an ergogenic
aid forsports performance in highly trained athletes: a critical
review. Int J Sports Med 18(7):491-496, 1997.
43. Kreider RB, et al: Effects of creatine supplementation on body
composition,strength, and sprint performance. Med Sci Sports
Exerc 30(1):73-82, 1998.
44. Balsom PD, et al: Creatine supplementation per se does not
enhance endurance exercise performance.Acta Physiol Scand
149(4):521-523, 1993.
45. Harris RC, Soderlund K, Hultman E: Elevation of creatine in
resting and exercised muscle of normal subjects by creatine
supplementation. Clin Sci (Lond) 83(3):367-374, 1992.
46. Clark JF: Creatine: A review of its nutritional applications in
sport. Nutrition 14(3):322-324, 1998.
47. Casey A, et al: Creatine ingestion favorably affects performance
and muscle metabolism during maximal exercise in
humans. Am J Physiol 271(1):E31-E37, 1996.
48. Volek JS: Creatine supplementation: its effect on human
muscular performance and body composition.J Strength Cond
Res 10:200-210, 1996.
49. Feldman EB: Creatine: a dietary supplement and ergogenic
aid. Nutr Rev 57(2):45-50, 1999.
50. Yarasheski KE: Growth hormone effects on metabolism, body
composition, muscle mass, and strength. Exerc Sport Sci Rev
22:285-312. 1994.
51. Risser WL: Sports medicine. Pediatr Rev 14(11):424-431,
1993.
52. Bidlingmaier M, Wu Z, Strasburger CJ: Doping with growth
hormone. J Pediatr Endocrinol Metab 14(8):1077-1083,
2001.
53. Jenkins PJ: Growth hormone and exercise: physiology, use and
abuse. Growth Horm IGF Res 11(Suppl A):S71-S77, 2001.
54. Rickert VI, et al: Human growth hormone: a new substance
of abuse among adolescents? Clin Pediatr (Phila) 31(12):723-
726, 1992.
55. Council Report: Drug abuse in athletes, anabolic steroids and
human growth hormone. J Am Med Assoc 259:1703-1705,
1988.
56. Berglund B, Ekblom B: Effect of recombinant human erythropoietin
treatment on blood pressure and some haematological
parameters in healthy men. J Intern Med 229(2):125-130,
1991.
57. Williams MH: Nutritional supplements for strength trained
athletes. Sports Sci Exchange 6:1-6, 1993.
58. Williams MH: Facts and fallacies of purported ergogenic
amino acid supplements. Clin Sports Med 18(3):633-649,
1999.
59. Vukovich MD, Dreifort GD: Effect of beta-hydroxy betamethylbutyrate
on the onset of blood lactate accumulation
and VO2 peak in endurance-trained cyclists. J Strength Cond
Res 15(4):491-497, 2001.
60. Knitter AE, et al: Effects of beta-hydroxy-beta-methylbutyrate
on muscle da
Post Disclaimer
Professional Scope of Practice *
The information on this blog site is not intended to replace a one-on-one relationship with a qualified healthcare professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those found on this site and our family practice-based chiromed.com site, focusing on restoring health naturally for patients of all ages.
Our areas of chiropractic practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is limited to chiropractic, musculoskeletal, physical medicine, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somato-visceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and their jurisdiction of licensure. We use functional health & wellness protocols to treat and support care for the injuries or disorders of the musculoskeletal system.
Our videos, posts, topics, subjects, and insights cover clinical matters and issues that relate to and directly or indirectly support our clinical scope of practice.*
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies available to regulatory boards and the public upon request.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: [email protected]
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License # TX5807
New Mexico DC License # NM-DC2182
Licensed as a Registered Nurse (RN*) in Texas & Multistate
Texas RN License # 1191402
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)
(Licensed Medical Doctor)
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933
Licenses and Board Certifications:
MD: Medical Doctor
DC: Doctor of Chiropractic
APRNP: Advanced Practice Registered Nurse
FNP-BC: Family Practice Specialization (Multi-State Board Certified)
RN: Registered Nurse (Multi-State Compact License)
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics
Memberships & Associations:
TCA: Texas Chiropractic Association: Member ID: 104311
AANP: American Association of Nurse Practitioners: Member ID: 2198960
ANA: American Nurse Association: Member ID: 06458222 (District TX01)
TNA: Texas Nurse Association: Member ID: 06458222
NPI: 1205907805
| Primary Taxonomy | Selected Taxonomy | State | License Number |
|---|---|---|---|
| No | 111N00000X - Chiropractor | NM | DC2182 |
| Yes | 111N00000X - Chiropractor | TX | DC5807 |
| Yes | 363LF0000X - Nurse Practitioner - Family | TX | 1191402 |
| Yes | 363LF0000X - Nurse Practitioner - Family | FL | 11043890 |
| Yes | 363LF0000X - Nurse Practitioner - Family | CO | C-APN.0105610-C-NP |
| Yes | 363LF0000X - Nurse Practitioner - Family | NY | N25929 |
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
(Board Certified: Family Practice Nurse Practitioner—Multistate)*
(Licensed Nurse Practitioner & Chiropractor - Multistate)*
Clinical Director
Digital Business Card
Dr. Maria Cardenas, MD
(Board Certified: Internal Medicine)*
(Licensed Medical Doctor)*
Medical Director, Clinical Director & Collaborative Physician
NPI # 1164426749
MD License #: J2933



 Anabolic-Androgenic Steroids
Anabolic-Androgenic Steroids
Comments are closed.